Non-contact temperature measurement for industry and R&D
Optris has been developing and manufacturing innovative infrared measurement devices for non-contact temperature measurement, including infrared cameras, and stationary industrial IR thermometers for area and point measurement, for more than 20 years.
Our comprehensive product portfolio comprises infrared measurement devices for different industrial applications as well as research & development. Along with our free thermal analysis software, our measurement devices enable constant monitoring and control of virtually every manufacturing process, and reductions in production costs through specific process optimization.
More Industries
INDUSTRY
INDUSTRY
INDUSTRY
INDUSTRY
INDUSTRY
INDUSTRY
INDUSTRY
INDUSTRY
INDUSTRY
The process and product temperature is an important physical indicator for manufacturing processes and ensures a high quality level of the production line.
All Optris products apply in different areas, covering the non-contact temperature measurement. This covers the plastic industry, the food industry as well as the solar industry and life science.
Infrared thermometers / pyrometers for







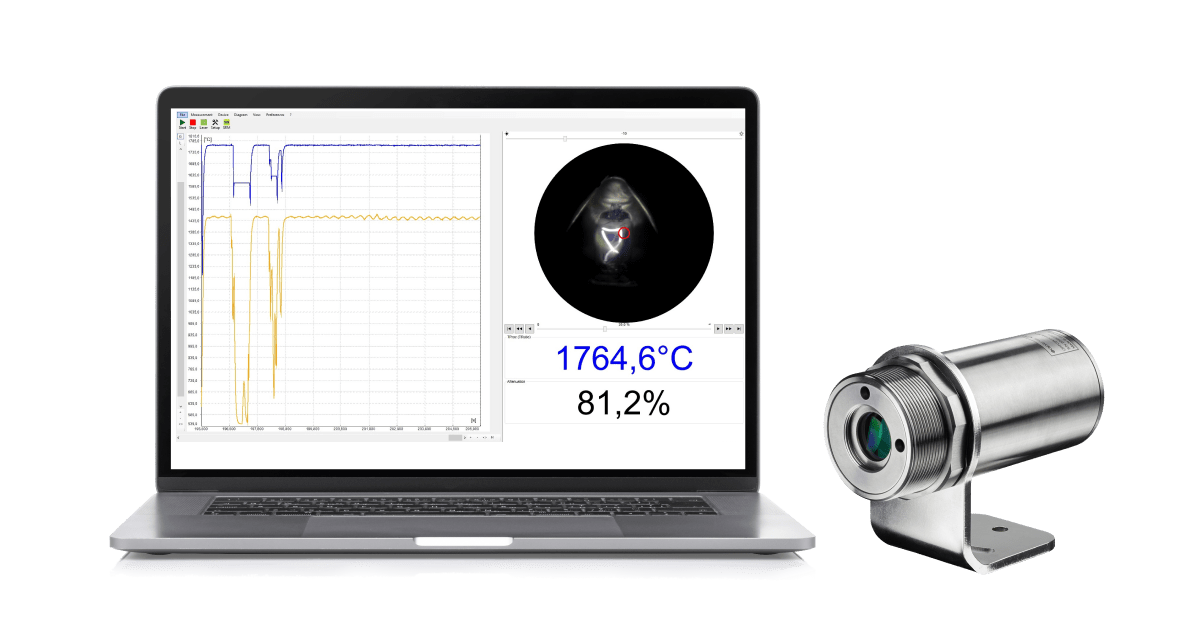
Optris infrared thermometers and pyrometers for spot measurements are particularly well suited for precise temperature monitoring of industrial manufacturing processes, research and development, and function checks of a diverse range of devices and systems.
Infrared Cameras / Thermal imagers for





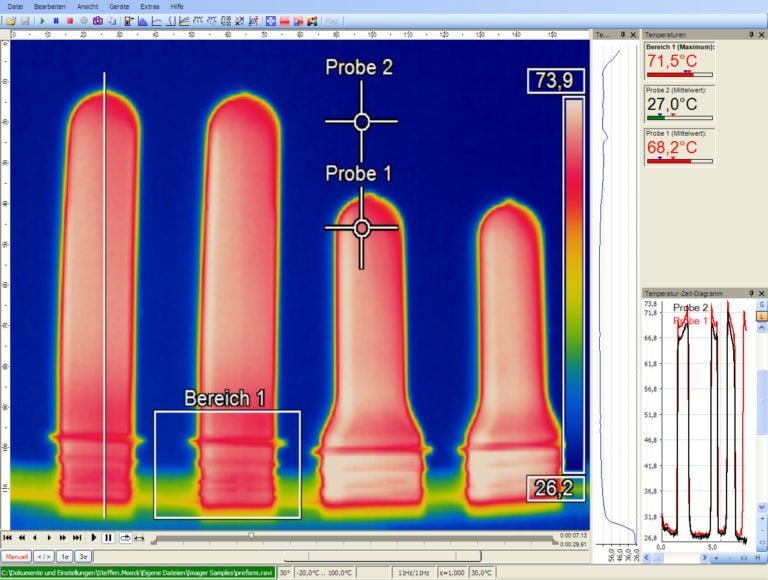
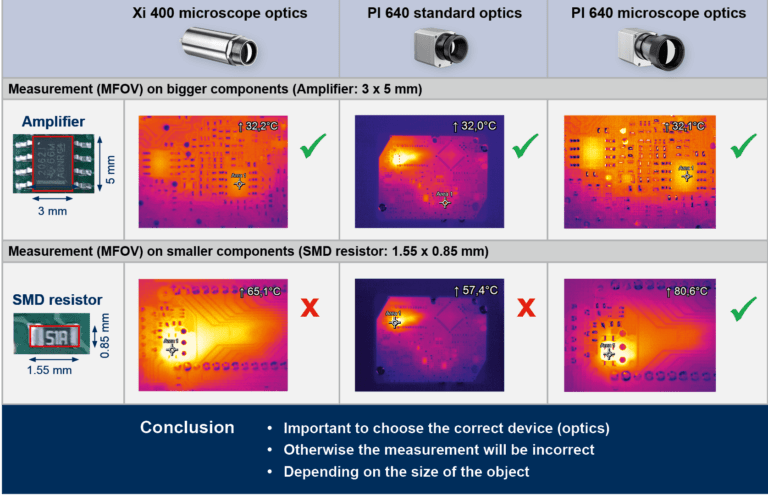
Optris’ infrared cameras are fully radiometric stationary thermographic systems with an excellent price-performance ratio. The thermal imaging cameras are connected to a PC via USB and they are immediately ready to be used. Temperature data is displayed through the license-free analysis software optris PIX Connect.
Upcoming Events
Event Type
All
Exhibition
IR Workshop
Webinar
Country
All
Austria
Belgium
Brazil
China
Czech Republic
Finland
Germany
India
Mexico
Poland
Saudi Arabia
Slovenia
Turkey
United Arab Emirates
United Kingdom
USA
Event Tag
All
2023
2023
2023
2023
2023
2023
2024
2024
2024
2024
2024
2024
Past and Future Events
All
Only Past Events
Only Future Events
Virtual Events
All
Virtual Events
Non Virtual Events